Michael Barbella, Managing Editor03.15.19
Christopher J. Centeno, M.D., could arguably be considered the “father” of regenerative medicine. He claims, after all, to be the first person on the planet to perform stem cell injections to treat joint arthritis and various other conditions.
There are several worthy contenders, including Arnie Caplan, Ph.D., pioneer of mesenchymal stem cell use (late 1980s); Philippe Henigou, developer of bone marrow concentrate treatment for bone disease (early 1990s); or Svante Gehring, M.D., the first physician to document use of platelet-rich plasma in ophthalmology (1999).
Centeno, however, believes the title should go to Ohio surgeon George S. Hackett, M.D., who clinically and scientifically demonstrated the efficacy of irritating tissue to promote healing back in 1963. Hackett’s studies showed that creating controlled inflammation could increase ligament size by up to 35-40 percent (more recent analyses have confirmed those initial findings).
While certainly revolutionary, Hackett’s work nevertheless was pre-dated by several thousand years: The concept of irritating tissue to promote healing actually originated with the ancient Greeks. Hippocrates reportedly treated Olympic javelin throwers with unstable shoulders by touching a “slender hot iron” to the joint’s ligaments (the heat would irritate the ligament, causing it to tighten).
Chronologically speaking, then, Hippocrates is deserving of the regenerative medicine “father” title. Yet Centeno maintains that Hackett warrants a place in history, if only for his contributions to the evolution of the field.
“So, is there one modern ‘father’?” Centeno, an international expert and regenerative medicine specialist, asked in a blog last spring. “You could make arguments for Hernigou, Caplan, the early users of PRP, and so on. However, if you had to award the title to one person, that honor likely goes to George Hackett, who figured out that you could help ligaments heal with injections.”
Regardless of its origins, regenerative medicine has become a fast-growing subsegment of the multi billion-dollar orthobiologics industry, thanks to an aging world population and advanced stem cell/gene therapy technologies in developed countries. ODT’s January/February feature “Natural Selection” examines the innovation being developed in this burgeoning sector that eventually will contribute to its double-digit annual growth over the next half-decade.
Ryanne Early, CEO of biological material-derived product manufacturer Seed Biotech Inc., shared her insights on the market for the story. Her full input is provided in the following Q&A.
Michael Barbella: Please discuss the current trends you see shaping the orthobiologics market.
Ryanne Early: The orthobiologics market is being influenced by an aging and more active population as well as an increasing rate of obesity. This change in demographics leads to an increased number of associated musculoskeletal injuries. The direct result of this is the requirement to develop cost-conscious orthobiologic products that are affordable, can be performed as outpatient procedures, and are easy to use. This will decrease overall medical costs and increase patients' ability to recover quickly. Building on several years of strong performance, orthobiologic products have come of age over the past several years, proving to be more effective in many instances at repairing injured or defective tissues than other types of medical devices. Continued success of well-known product lines has led surgeons to realize the potential of a wide range of biologic products available for accelerating tissue healing. The continuous improvement in the safety and supply of tissue products and continued innovation in orthobiological products will be necessary to address these needs.
Barbella: What factors are driving innovation in the orthobiologics market?
Early: A variety of factors are driving innovation in the orthobiologics market. Primarily, the landscape of regulatory approval drives basic and clinical research and development costs. Indeed, creating products that bring value to procedures across multiple markets, have multi-use properties, and are easy to use has become a current trend. It is expected that products developed today will be applied or used in the established trend of minimally invasive procedures (laser, robotics), providing limited to no severe side effects (from clinical tests).
Another innovation-driving factor is the improvement in patient pain management and an increase in healing times due to co-morbidities. Cost is also a big factor in determining innovation in this market, with the goal to reduce hospital stay times and decrease rehabilitation. Physicians and patients are becoming more knowledgeable, which in turn, requires device manufacturers to provide them with innovative products. R&D investments have increased, along with expansion of biotechnology companies, driven by market need and competition. These budgets are allocated to product research and development, boosting innovation.
Barbella: What factors are impeding growth in the orthobiologics market? How can these challenges be overcome?
Early: The biggest growth-impeding factor in the orthobiologics market is the ability to get reimbursement for a product. Another major factor impeding growth in this industry is the cost associated with the regulatory pathway most products have to follow to get on the market. Additionally, limited budgets lead to limited competition for improved products, so smaller companies have a harder time competing and growing against the big players. Finally, there is a lack of empirical/definitive/accurate data as to the clinical efficacy of the orthobiologic product. Clearly defined pathways to reimbursement and proof of clinical efficacy are needed for product growth.
Barbella: What new technologies are in the works?
Early: Seed Biotech is currently working on alternative sources and unconventional solutions to create novel orthobiologics products. We are focusing on more biologic-based, renewable products for non-homologous uses. Additionally, modifications to existing products are underway to improve consistency of the handling and biologic reactions with less risk of side effects. Finally, additional testing of alternate manufacturing techniques to produce novel product forms is underway to provide increased yield as well as expanded applications to fill unmet medical needs.
Seed Biotech's innovative technologies are based on a proprietary processing method which retains more of the bioactivity of the material being used. This is being achieved with the scientific and technical expertise of Seed Biotech by utilizing higher quality scientific evidence, advanced technology, and superior manufacturing as compared to current products on the market.
Barbella: Where does innovation come from in the orthobiologics market? How does Seed Biotech stay innovative amid all the competition?
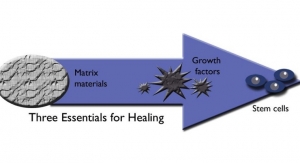
Early: At Seed Biotech, we use precise methods to obtain measurable results to create better biologics. Innovation is driven by the market trending towards the use of biologics for tissue regeneration. Biological materials provide a variety of inductive functions and are naturally regenerative, providing either an active process for tissue regeneration or a scaffold for healing following surgery. These products are based on natural biologics and scaffolds that the body can recognize and utilize with its normal healing pathways to repair and/or remodel injured tissues.
Barbella: How does the regulatory landscape for orthobiologics differ from other sectors of the orthopedic market, and how does this impact product innovation?
Early: As with all products, for all sectors of biotechnology markets, the regulatory pathway is determined by a product's base material, manufacturing methods, and intended use. In the orthobiologics market, there are a variety of regulatory pathways depending on the product:
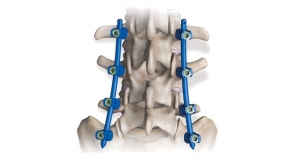
Barbella: What is the dominant orthobiologics application—spinal fusion, trauma repair, reconstructive surgery? What factors are driving growth in this particular application?
Early: In the current market, orthobiologics has proven to be a contributing factor in the success of the majority of orthopedic and spinal surgical procedures, and are therefore more widely used and accepted. Spinal fusion is the dominant procedure utilizing orthobiologics. In spinal surgery applications, improvements and new products are accepted and favored, and reimbursement continues. Developing new or improved orthobiologics that complement the current and highest use procedures will provide a more expected and confident return on investments. In return, more money is put towards research and development to create better, safer, and more cost-conscious orthobiologics products.
There are several worthy contenders, including Arnie Caplan, Ph.D., pioneer of mesenchymal stem cell use (late 1980s); Philippe Henigou, developer of bone marrow concentrate treatment for bone disease (early 1990s); or Svante Gehring, M.D., the first physician to document use of platelet-rich plasma in ophthalmology (1999).
Centeno, however, believes the title should go to Ohio surgeon George S. Hackett, M.D., who clinically and scientifically demonstrated the efficacy of irritating tissue to promote healing back in 1963. Hackett’s studies showed that creating controlled inflammation could increase ligament size by up to 35-40 percent (more recent analyses have confirmed those initial findings).
While certainly revolutionary, Hackett’s work nevertheless was pre-dated by several thousand years: The concept of irritating tissue to promote healing actually originated with the ancient Greeks. Hippocrates reportedly treated Olympic javelin throwers with unstable shoulders by touching a “slender hot iron” to the joint’s ligaments (the heat would irritate the ligament, causing it to tighten).
Chronologically speaking, then, Hippocrates is deserving of the regenerative medicine “father” title. Yet Centeno maintains that Hackett warrants a place in history, if only for his contributions to the evolution of the field.
“So, is there one modern ‘father’?” Centeno, an international expert and regenerative medicine specialist, asked in a blog last spring. “You could make arguments for Hernigou, Caplan, the early users of PRP, and so on. However, if you had to award the title to one person, that honor likely goes to George Hackett, who figured out that you could help ligaments heal with injections.”
Regardless of its origins, regenerative medicine has become a fast-growing subsegment of the multi billion-dollar orthobiologics industry, thanks to an aging world population and advanced stem cell/gene therapy technologies in developed countries. ODT’s January/February feature “Natural Selection” examines the innovation being developed in this burgeoning sector that eventually will contribute to its double-digit annual growth over the next half-decade.
Ryanne Early, CEO of biological material-derived product manufacturer Seed Biotech Inc., shared her insights on the market for the story. Her full input is provided in the following Q&A.
Michael Barbella: Please discuss the current trends you see shaping the orthobiologics market.
Ryanne Early: The orthobiologics market is being influenced by an aging and more active population as well as an increasing rate of obesity. This change in demographics leads to an increased number of associated musculoskeletal injuries. The direct result of this is the requirement to develop cost-conscious orthobiologic products that are affordable, can be performed as outpatient procedures, and are easy to use. This will decrease overall medical costs and increase patients' ability to recover quickly. Building on several years of strong performance, orthobiologic products have come of age over the past several years, proving to be more effective in many instances at repairing injured or defective tissues than other types of medical devices. Continued success of well-known product lines has led surgeons to realize the potential of a wide range of biologic products available for accelerating tissue healing. The continuous improvement in the safety and supply of tissue products and continued innovation in orthobiological products will be necessary to address these needs.
Barbella: What factors are driving innovation in the orthobiologics market?
Early: A variety of factors are driving innovation in the orthobiologics market. Primarily, the landscape of regulatory approval drives basic and clinical research and development costs. Indeed, creating products that bring value to procedures across multiple markets, have multi-use properties, and are easy to use has become a current trend. It is expected that products developed today will be applied or used in the established trend of minimally invasive procedures (laser, robotics), providing limited to no severe side effects (from clinical tests).
Another innovation-driving factor is the improvement in patient pain management and an increase in healing times due to co-morbidities. Cost is also a big factor in determining innovation in this market, with the goal to reduce hospital stay times and decrease rehabilitation. Physicians and patients are becoming more knowledgeable, which in turn, requires device manufacturers to provide them with innovative products. R&D investments have increased, along with expansion of biotechnology companies, driven by market need and competition. These budgets are allocated to product research and development, boosting innovation.
Barbella: What factors are impeding growth in the orthobiologics market? How can these challenges be overcome?
Early: The biggest growth-impeding factor in the orthobiologics market is the ability to get reimbursement for a product. Another major factor impeding growth in this industry is the cost associated with the regulatory pathway most products have to follow to get on the market. Additionally, limited budgets lead to limited competition for improved products, so smaller companies have a harder time competing and growing against the big players. Finally, there is a lack of empirical/definitive/accurate data as to the clinical efficacy of the orthobiologic product. Clearly defined pathways to reimbursement and proof of clinical efficacy are needed for product growth.
Barbella: What new technologies are in the works?
Early: Seed Biotech is currently working on alternative sources and unconventional solutions to create novel orthobiologics products. We are focusing on more biologic-based, renewable products for non-homologous uses. Additionally, modifications to existing products are underway to improve consistency of the handling and biologic reactions with less risk of side effects. Finally, additional testing of alternate manufacturing techniques to produce novel product forms is underway to provide increased yield as well as expanded applications to fill unmet medical needs.
Seed Biotech's innovative technologies are based on a proprietary processing method which retains more of the bioactivity of the material being used. This is being achieved with the scientific and technical expertise of Seed Biotech by utilizing higher quality scientific evidence, advanced technology, and superior manufacturing as compared to current products on the market.
Barbella: Where does innovation come from in the orthobiologics market? How does Seed Biotech stay innovative amid all the competition?
Early: At Seed Biotech, we use precise methods to obtain measurable results to create better biologics. Innovation is driven by the market trending towards the use of biologics for tissue regeneration. Biological materials provide a variety of inductive functions and are naturally regenerative, providing either an active process for tissue regeneration or a scaffold for healing following surgery. These products are based on natural biologics and scaffolds that the body can recognize and utilize with its normal healing pathways to repair and/or remodel injured tissues.
Barbella: How does the regulatory landscape for orthobiologics differ from other sectors of the orthopedic market, and how does this impact product innovation?
Early: As with all products, for all sectors of biotechnology markets, the regulatory pathway is determined by a product's base material, manufacturing methods, and intended use. In the orthobiologics market, there are a variety of regulatory pathways depending on the product:
- HCTP: Allografts (361)
- Medical devices require either a 510(k) or IDE (investigational device exemption) and need pre-market approval (PMA)
- Biologic: BLA/IND (351) PMA
- Drug: PMA
- Combination device PMA
Barbella: What is the dominant orthobiologics application—spinal fusion, trauma repair, reconstructive surgery? What factors are driving growth in this particular application?
Early: In the current market, orthobiologics has proven to be a contributing factor in the success of the majority of orthopedic and spinal surgical procedures, and are therefore more widely used and accepted. Spinal fusion is the dominant procedure utilizing orthobiologics. In spinal surgery applications, improvements and new products are accepted and favored, and reimbursement continues. Developing new or improved orthobiologics that complement the current and highest use procedures will provide a more expected and confident return on investments. In return, more money is put towards research and development to create better, safer, and more cost-conscious orthobiologics products.