Sam Brusco, Associate Editor04.02.19
Neurostimulation therapy (targeted electrical pulses to manage chronic pain) comes in two major forms: spinal cord stimulation (SCS) and peripheral nerve stimulation (PNS). The latter of the two was invented in the 1960s and involve surgery to implant a small electrical device next to a peripheral nerve—those located beyond the brain or spinal cord. It is used to treat certain types of complex regional pain syndrome and pain due to peripheral nerve injuries, as well as back pain, migraines, and potentially urinary bladder incontinence.
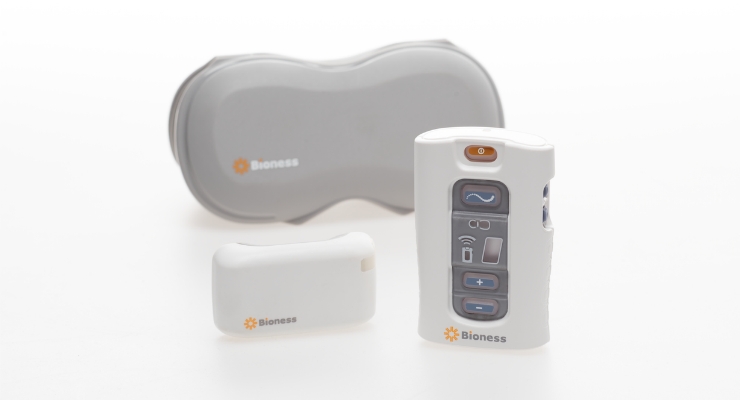
Bioness, a Valencia, Calif.-based medical device company that produces implantable and external neuromodulation systems, robotic systems, and software-based therapy programs providing functional and therapeutic benefits for those affected by pain, central nervous system disorders, and orthopedic injuries, launched its StimRouter device in 2015. StimRouter uses PNS therapy to treat intractable pain originating in a peripheral nerve.
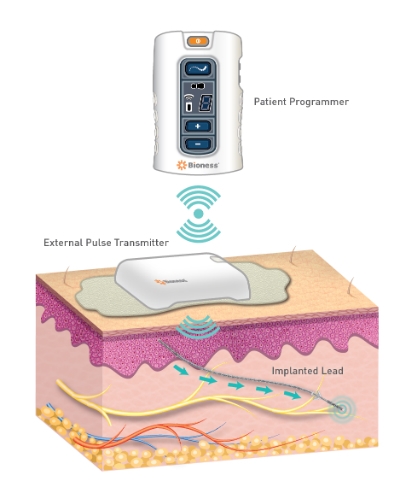
Applications for the technology include chronic pain conditions located at or related to the upper and lower limbs, entrapment syndromes, intercostal neuralgias, or other peripheral injuries or diseases. The StimRouter implant consists of a passive, 15 cm lead with electrodes at one end and a receiver at the other that is implanted percutaneously with a small incision under local anesthesia. The lead’s tip is placed near the peripheral nerve causing the chronic pain. A Patient Programmer resembling a small remote control controls the duration and intensity of electrical stimulation.
To gain more insight into how technology can help to curb or eliminate opioid use, I recently spoke with Mark Geiger, Bioness’ global director of marketing, implantables. Some of his input is included in the pain management-focused cover story for the March/April issue of ODT, entitled “Pain, Pain, Go Away.” The full extent of Mark’s comments are featured here.
Sam Brusco: Why is it important for medical technology to be part of a pain management regimen?
Mark Geiger: Medical technology is an important part of a pain management regimen because the technology, specific to neuromodulation, has progressed to provide effective, safe, non-addictive, reversible, and long-term solutions for chronic pain treatment. Many neuromodulation devices, including peripheral nerve stimulation (PNS) products like the StimRouter, are minimally invasive and only require a 15-30 minute implant by a doctor while the patient is awake under local anesthesia. These devices can put the patient in charge of their own pain management at home.
Additionally, a recent study presented in a poster session at the North American Neuromodulation Society shows PNS technology helps patients reduce or eliminate their reliance on opioids as a treatment and can significantly reduce their pain levels enough to move forward with their lives.1
Brusco: How does the StimRouter device itself work? How does the accompanying patient programmer enhance the therapy?
The StimRouter System is designed to accommodate patients who have an active lifestyle and to empower them with complete control of their pain management treatment.
Geiger: The StimRouter implant is a passive, 15cm lead with electrodes at one end and a receiver on the back end. An External Pulse Transmitter or “patch” is worn on the skin over the implant to power it and send the chosen stimulation signal to the implant.
The system also comes with a “Patient Programmer” resembling a small remote control the patient uses to turn the system on, off, up, and down to control their chronic pain. The patient programmer also has a display that allows them to monitor and manage their stimulation programs and level of stimulation intensity.
Brusco: How does StimRouter differ from other neurostimulation therapies for chronic pain?
Geiger: The StimRouter was the first system of its kind designed specifically to treat focal, chronic pain of a peripheral nerve origin. When compared to the more established spinal cord stimulation systems offered by several large medical device corporations (which involves major surgery with a large, global implant), the StimRouter has the electronics (processor, battery) residing on the wearable outside the body, resulting in a very small, simple implant process while the patient is awake. This eliminates many of the risks typically associated with a spinal cord stimulator, such as implant migration.
Brusco: Are you finding that clinicians, patients, and payers are embracing medical technology as a solution to manage pain?
Geiger: Healthcare professionals are becoming more interested in medical technology to better manage chronic pain, as it not only provides a non-opioid solution, but clinical data is showing the positive impact devices like the StimRouter can have on a patient’s quality of life.
Brusco: If so, what do you believe convinced them to use the technology over traditional pain management methods?
Geiger: As with most new medical technologies, clinicians begin by using it as a last resort when the established treatment algorithm has been exhausted. As clinicians become more comfortable with it and prove to themselves and their patients that it works, they move the product’s use upstream in front of other products/treatments. With the StimRouter, we’ve seen this change in the treatment algorithm at large pain centers all over the U.S., putting it ahead of the more invasive spinal cord stimulation products and destructive nerve ablation procedures. The data speaks for itself. For example, a recent poster from the North American Neuromodulation Society (Jan, 2019) showed an average of 71 percent pain reduction across many different peripheral nerves that were stimulated in the study. In addition, 94 percent of the patients had at least a 50 percent reduction in opioid use.
Clinical data featured in another poster shows the implications of PNS technology to the US healthcare system: an 81 percent reduction in cost and a 53 percent reduction in time to effective therapy.
Brusco: If not, what do you believe is necessary to change their opinions?
Geiger: For those hesitant to offer medical technology, one of the biggest impediments to the adoption of new technology like peripheral nerve stimulators is the reimbursement system. While the reimbursement for peripheral nerve stimulation (PNS) has improved over the past 2+ years, there is a large disparity between facility and physician payments for PNS compared to more established therapies. PNS can thrive when this gap is removed or thrive where it doesn’t exist as with large academic institutions.
The other impediment to technology adoption is clinical data. The StimRouter is the only PNS system backed by a multi-center, randomized, controlled trial (RCT) that met all FDA requirements to justify FDA clearance. In addition to meeting the target FDA endpoint, the following highlights from the study were reported:
No serious adverse events including migration and infection.
In addition to the first and only RCT for PNS, we have started the first and only PNS Registry, a five-year, 30 center, 1,000 patient registry to further study the optimal applications and best practices associated with the StimRouter therapy.
Brusco: What plans are in the future for StimRouter and/or other Bioness pain management technologies?
Geiger: The StimRouter PNS System is truly a platform technology. In addition to the chronic pain application, We are invested in an Investigational Device Exemption (IDE) study in the U.S. for the treatment of overactive bladder (OAB) through the stimulation of the tibial nerve. OAB affects approximately 33 million people in the U.S. and 526 million people worldwide, so the opportunity to help these patients through the StimRouter is tremendous. Other potential applications are being investigated as well.
Reference
1 Chakravarthy, M.D., Ph.D., Krishnan & Oswald, M.D., Ph.D., Jessica. (2017). A case series on the use of peripheral nerve stimulation for focal mono-neuropathy treatment. San Diego, CA. University of California, San Diego.
Bioness, a Valencia, Calif.-based medical device company that produces implantable and external neuromodulation systems, robotic systems, and software-based therapy programs providing functional and therapeutic benefits for those affected by pain, central nervous system disorders, and orthopedic injuries, launched its StimRouter device in 2015. StimRouter uses PNS therapy to treat intractable pain originating in a peripheral nerve.
Applications for the technology include chronic pain conditions located at or related to the upper and lower limbs, entrapment syndromes, intercostal neuralgias, or other peripheral injuries or diseases. The StimRouter implant consists of a passive, 15 cm lead with electrodes at one end and a receiver at the other that is implanted percutaneously with a small incision under local anesthesia. The lead’s tip is placed near the peripheral nerve causing the chronic pain. A Patient Programmer resembling a small remote control controls the duration and intensity of electrical stimulation.
To gain more insight into how technology can help to curb or eliminate opioid use, I recently spoke with Mark Geiger, Bioness’ global director of marketing, implantables. Some of his input is included in the pain management-focused cover story for the March/April issue of ODT, entitled “Pain, Pain, Go Away.” The full extent of Mark’s comments are featured here.
Sam Brusco: Why is it important for medical technology to be part of a pain management regimen?
Mark Geiger: Medical technology is an important part of a pain management regimen because the technology, specific to neuromodulation, has progressed to provide effective, safe, non-addictive, reversible, and long-term solutions for chronic pain treatment. Many neuromodulation devices, including peripheral nerve stimulation (PNS) products like the StimRouter, are minimally invasive and only require a 15-30 minute implant by a doctor while the patient is awake under local anesthesia. These devices can put the patient in charge of their own pain management at home.
Additionally, a recent study presented in a poster session at the North American Neuromodulation Society shows PNS technology helps patients reduce or eliminate their reliance on opioids as a treatment and can significantly reduce their pain levels enough to move forward with their lives.1
Brusco: How does the StimRouter device itself work? How does the accompanying patient programmer enhance the therapy?

The StimRouter System is designed to accommodate patients who have an active lifestyle and to empower them with complete control of their pain management treatment.
The system also comes with a “Patient Programmer” resembling a small remote control the patient uses to turn the system on, off, up, and down to control their chronic pain. The patient programmer also has a display that allows them to monitor and manage their stimulation programs and level of stimulation intensity.
Brusco: How does StimRouter differ from other neurostimulation therapies for chronic pain?
Geiger: The StimRouter was the first system of its kind designed specifically to treat focal, chronic pain of a peripheral nerve origin. When compared to the more established spinal cord stimulation systems offered by several large medical device corporations (which involves major surgery with a large, global implant), the StimRouter has the electronics (processor, battery) residing on the wearable outside the body, resulting in a very small, simple implant process while the patient is awake. This eliminates many of the risks typically associated with a spinal cord stimulator, such as implant migration.
Brusco: Are you finding that clinicians, patients, and payers are embracing medical technology as a solution to manage pain?
Geiger: Healthcare professionals are becoming more interested in medical technology to better manage chronic pain, as it not only provides a non-opioid solution, but clinical data is showing the positive impact devices like the StimRouter can have on a patient’s quality of life.
Brusco: If so, what do you believe convinced them to use the technology over traditional pain management methods?
Geiger: As with most new medical technologies, clinicians begin by using it as a last resort when the established treatment algorithm has been exhausted. As clinicians become more comfortable with it and prove to themselves and their patients that it works, they move the product’s use upstream in front of other products/treatments. With the StimRouter, we’ve seen this change in the treatment algorithm at large pain centers all over the U.S., putting it ahead of the more invasive spinal cord stimulation products and destructive nerve ablation procedures. The data speaks for itself. For example, a recent poster from the North American Neuromodulation Society (Jan, 2019) showed an average of 71 percent pain reduction across many different peripheral nerves that were stimulated in the study. In addition, 94 percent of the patients had at least a 50 percent reduction in opioid use.
Clinical data featured in another poster shows the implications of PNS technology to the US healthcare system: an 81 percent reduction in cost and a 53 percent reduction in time to effective therapy.
Brusco: If not, what do you believe is necessary to change their opinions?
Geiger: For those hesitant to offer medical technology, one of the biggest impediments to the adoption of new technology like peripheral nerve stimulators is the reimbursement system. While the reimbursement for peripheral nerve stimulation (PNS) has improved over the past 2+ years, there is a large disparity between facility and physician payments for PNS compared to more established therapies. PNS can thrive when this gap is removed or thrive where it doesn’t exist as with large academic institutions.
The other impediment to technology adoption is clinical data. The StimRouter is the only PNS system backed by a multi-center, randomized, controlled trial (RCT) that met all FDA requirements to justify FDA clearance. In addition to meeting the target FDA endpoint, the following highlights from the study were reported:
No serious adverse events including migration and infection.
- 50 percent of the treatment group rated their satisfaction at 8 or higher on a 10-point scale.
- 53 percent of the treatment group rated their global impression of change in activity limitations, symptoms, emotions and overall quality of life related to their painful condition between 5-7 on a 7-point scale.
- 31 percent of the treatment group rated their satisfaction at a 10 on a 10-point scale.
In addition to the first and only RCT for PNS, we have started the first and only PNS Registry, a five-year, 30 center, 1,000 patient registry to further study the optimal applications and best practices associated with the StimRouter therapy.
Brusco: What plans are in the future for StimRouter and/or other Bioness pain management technologies?
Geiger: The StimRouter PNS System is truly a platform technology. In addition to the chronic pain application, We are invested in an Investigational Device Exemption (IDE) study in the U.S. for the treatment of overactive bladder (OAB) through the stimulation of the tibial nerve. OAB affects approximately 33 million people in the U.S. and 526 million people worldwide, so the opportunity to help these patients through the StimRouter is tremendous. Other potential applications are being investigated as well.
Reference
1 Chakravarthy, M.D., Ph.D., Krishnan & Oswald, M.D., Ph.D., Jessica. (2017). A case series on the use of peripheral nerve stimulation for focal mono-neuropathy treatment. San Diego, CA. University of California, San Diego.