Sean Fenske, Editor11.21.17
Most sports fans are familiar with the phrase “bulletin board material.” It seems to be a favorite topic for sports reporters to bring up with coaches and players when someone from the opposing team makes derogatory comments. Those comments make newspaper headlines and thus, become “bulletin board material.” A coach might use such headlines or comments to provide a little extra motivation for his players to dig just a bit deeper on game day.
Well, it seems such material has made its way into the orthopedic technology space. More specifically, one might say it’s emerged in the ongoing PEEK vs. titanium debate with regard to material of choice for spinal implants. The PEEK folks point to the issue of artifacts appearing in X-rays that is associated with titanium. The titanium contingent, on the other hand, brings up the problem of fibrous tissue surrounding PEEK implants and adversely impacting integration with surrounding bone. I’ve discussed this debate previously (see my Editor’s Letter in the March/April 2017 issue).
Having recently attended the annual NASS meeting in October, this debate was thrust to the forefront of my attention once more. This time, however, there was a bit of a difference. Some companies were actively responding to the other sides’ criticisms of their material technology. Really, it was primarily the PEEK-based companies who were addressing the issue of the fibrous tissue formation.
The first company to respond (that I encountered) to the “bulletin board material” being pitched to the industry by the titanium faction was Vertera Spine. I just shared my experience of first meeting the company’s CEO, Dr. Chris Lee, at the 2016 NASS meeting in last issue’s Editor’s Letter (Porous PEEK in the Limelight). For those who missed it, NuVasive bought the company a few months ago, so I wasn’t the only one who noticed the company’s innovative response to the concerns over PEEK-based implants.
Only a year later at the 2017 NASS meeting, I met with another company claiming to have a solution to the fibrous tissue concerns attributed to PEEK-based spinal implants. Austin, Texas-based DiFusion Technologies was founded in 2008 to focus on reducing the incidence of infections in orthopedic and spine surgery through the development of antimicrobial-based implants. In the process of their material experimentation, however, they stumbled upon a potential breakthrough that could significantly tip the scales in the PEEK vs. titanium debate.
“It was sort of a penicillin moment,” explained Derrick Johns, CEO of DiFusion Technologies. “We started out engineering antimicrobial polymers by first loading zeolite particles with silver before compounding them. But we discovered if we took the silver cations out of the zeolite, they imbued PEEK with a negative charge. Osteoblast cells are attracted to the negatively charged surface at a far higher rate than titanium, and yet, we were able to preserve the polymer’s outstanding visualization, modulus, and strength benefits.”
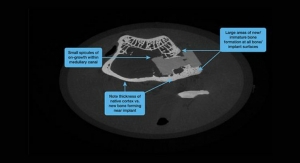
Essentially, the company figured out a way to get bone to interface with PEEK without a fibrous layer forming between the two. The very advantages of PEEK that make it such an attractive alternative to titanium were preserved, while the material’s negative aspect was eliminated. The company is now in the final stages of its FDA 510(k) approval process and expects the technology to be commercially available in the U.S. in early 2018.
Conversely, the primary issue brought up with regard to the use of titanium for spinal implants—the imaging artifacts—is actually a concern being addressed by companies who may not even produce implants. Rather, imaging technology companies are working on software and algorithm solutions to reduce this concern. Frankly, I’m not entirely sure there’s anything the titanium implant companies can do directly to address this issue other than to work with those imaging system manufacturers to determine if implant design alterations have any significant impact on the visualization challenges.
During my discussion with Johns at NASS, as well as with a representative from DiFusion’s PEEK provider (Solvay), we further explored the material solution they had discovered, in addition to other PEEK-based technology offerings, such as the one from Vertera Spine. It wasn’t very long before we came to the realization that a combination of the two could be an ideal answer. Take the porous surface texture created from Vertera’s process and use it on DiFusion Technologies’ negatively charged PEEK implant.
Ultimately, however, the hope would presumably be that NuVasive takes notice of this since the Vertera Spine process is now the property of the orthopedic OEM. Given the prevalence of M&A in today’s medical device space, however, it’s possible that NuVasive puts itself into position to leverage both technologies under one roof. That would certainly add an interesting new layer to the debate.
Well, it seems such material has made its way into the orthopedic technology space. More specifically, one might say it’s emerged in the ongoing PEEK vs. titanium debate with regard to material of choice for spinal implants. The PEEK folks point to the issue of artifacts appearing in X-rays that is associated with titanium. The titanium contingent, on the other hand, brings up the problem of fibrous tissue surrounding PEEK implants and adversely impacting integration with surrounding bone. I’ve discussed this debate previously (see my Editor’s Letter in the March/April 2017 issue).
Having recently attended the annual NASS meeting in October, this debate was thrust to the forefront of my attention once more. This time, however, there was a bit of a difference. Some companies were actively responding to the other sides’ criticisms of their material technology. Really, it was primarily the PEEK-based companies who were addressing the issue of the fibrous tissue formation.
The first company to respond (that I encountered) to the “bulletin board material” being pitched to the industry by the titanium faction was Vertera Spine. I just shared my experience of first meeting the company’s CEO, Dr. Chris Lee, at the 2016 NASS meeting in last issue’s Editor’s Letter (Porous PEEK in the Limelight). For those who missed it, NuVasive bought the company a few months ago, so I wasn’t the only one who noticed the company’s innovative response to the concerns over PEEK-based implants.
Only a year later at the 2017 NASS meeting, I met with another company claiming to have a solution to the fibrous tissue concerns attributed to PEEK-based spinal implants. Austin, Texas-based DiFusion Technologies was founded in 2008 to focus on reducing the incidence of infections in orthopedic and spine surgery through the development of antimicrobial-based implants. In the process of their material experimentation, however, they stumbled upon a potential breakthrough that could significantly tip the scales in the PEEK vs. titanium debate.
“It was sort of a penicillin moment,” explained Derrick Johns, CEO of DiFusion Technologies. “We started out engineering antimicrobial polymers by first loading zeolite particles with silver before compounding them. But we discovered if we took the silver cations out of the zeolite, they imbued PEEK with a negative charge. Osteoblast cells are attracted to the negatively charged surface at a far higher rate than titanium, and yet, we were able to preserve the polymer’s outstanding visualization, modulus, and strength benefits.”
Essentially, the company figured out a way to get bone to interface with PEEK without a fibrous layer forming between the two. The very advantages of PEEK that make it such an attractive alternative to titanium were preserved, while the material’s negative aspect was eliminated. The company is now in the final stages of its FDA 510(k) approval process and expects the technology to be commercially available in the U.S. in early 2018.
Conversely, the primary issue brought up with regard to the use of titanium for spinal implants—the imaging artifacts—is actually a concern being addressed by companies who may not even produce implants. Rather, imaging technology companies are working on software and algorithm solutions to reduce this concern. Frankly, I’m not entirely sure there’s anything the titanium implant companies can do directly to address this issue other than to work with those imaging system manufacturers to determine if implant design alterations have any significant impact on the visualization challenges.
During my discussion with Johns at NASS, as well as with a representative from DiFusion’s PEEK provider (Solvay), we further explored the material solution they had discovered, in addition to other PEEK-based technology offerings, such as the one from Vertera Spine. It wasn’t very long before we came to the realization that a combination of the two could be an ideal answer. Take the porous surface texture created from Vertera’s process and use it on DiFusion Technologies’ negatively charged PEEK implant.
Ultimately, however, the hope would presumably be that NuVasive takes notice of this since the Vertera Spine process is now the property of the orthopedic OEM. Given the prevalence of M&A in today’s medical device space, however, it’s possible that NuVasive puts itself into position to leverage both technologies under one roof. That would certainly add an interesting new layer to the debate.