GlobeNewswire06.14.18
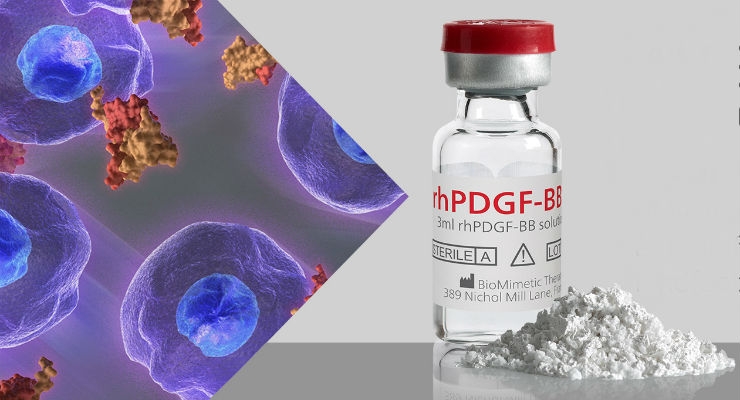
Wright Medical Group N.V. announced that it has received premarket approval (PMA) from the U.S. Food and Drug Administration (FDA) for AUGMENT Injectable Bone Graft for the same clinical indications as AUGMENT Bone Graft. AUGMENT Injectable is a combination product consisting of recombinant human platelet derived growth factor (rhPDGF-BB) and a blend of Type I collagen and Beta tri-calcium phosphate, which provides a clinically proven and safe and effective alternative to autograft for use in hindfoot and ankle fusion in an easy to use flowable formulation.
Robert Palmisano, president and chief executive officer, stated, “Building on the commercial and clinical success of AUGMENT Injectable in the Canadian and Australian markets, this important milestone allows Wright to expand our proven AUGMENT franchise and meet the needs of foot and ankle surgeons in the U.S. with a safe and effective alternative to autograft that includes significantly improved handling characteristics to enable easier and more precise delivery to the fusion site. In addition to the significantly improved handling characteristics with AUGMENT Injectable, one of the key differentiators continues to be the strength of the clinical evidence. Our approved PMA supplement for AUGMENT Injectable builds upon the existing body of evidence supporting the safety and effectiveness of AUGMENT Bone Graft and leverages clinical data from the U.S. and Canada for the injectable formulation.”
The U.S. launch of AUGMENT Injectable is expected to deliver sustained growth in the U.S. for the AUGMENT Regenerative Solutions platform over the next several years with a gradual ramp as the injectable form is reviewed by hospital value analysis committees and physician education programs are executed. The company will immediately start the process of finalizing its packaging carrying the FDA approved labeling and expects to begin selling product in the U.S. shortly and complete the rollout in the next four to six weeks.
Dr. Timothy Daniels, Chief of the Division of Orthopaedic Surgery of St. Michael’s Hospital and Professor at the University of Toronto in the Department of Surgery, commented, “In Canada, my colleagues and I have been using AUGMENT Injectable since 2011 based upon its ideal handling characteristics and impressive clinical results. FDA approval of AUGMENT Injectable provides a valuable enhancement to the existing AUGMENT product line for U.S. foot and ankle surgeons as a front-line biologic alternative that drives the biological healing process and eliminates the costs and clinical impacts of autograft harvesting.”
Dr. Jeremy McCormick, Associate Professor of Orthopaedic Surgery at Washington University School of Medicine in St. Louis, Mo., added, “I adopted AUGMENT Bone Graft shortly after it launched as a clinically proven tool that has improved patient outcomes. As my confidence in the product has grown, so too has my use of AUGMENT. Now, with the introduction of the new injectable version, delivery of the product to the fusion site will be simplified and more consistent, helping foot and ankle specialists use it more dependably in patients.”
AUGMENT Injectable is the first clinically proven injectable protein therapeutic to come to the U.S. orthopaedic market offering an alternative to autograft in ankle and/or hindfoot fusion procedures, which translates into an estimated market opportunity of approximately $300 million in the U.S. As FDA-approved alternatives to autograft in ankle and/or hindfoot fusion procedures, AUGMENT Bone Graft and AUGMENT Injectable offer a clear patient benefit by avoiding secondary surgical sites for the harvest of autograft tissue, which can result in prolonged harvest site pain and other comorbidities in some patients.
As a result of this approval, the company plans to update its 2018 annual guidance on its second quarter earnings call scheduled for August 8, 2018.
Robert Palmisano, president and chief executive officer, stated, “Building on the commercial and clinical success of AUGMENT Injectable in the Canadian and Australian markets, this important milestone allows Wright to expand our proven AUGMENT franchise and meet the needs of foot and ankle surgeons in the U.S. with a safe and effective alternative to autograft that includes significantly improved handling characteristics to enable easier and more precise delivery to the fusion site. In addition to the significantly improved handling characteristics with AUGMENT Injectable, one of the key differentiators continues to be the strength of the clinical evidence. Our approved PMA supplement for AUGMENT Injectable builds upon the existing body of evidence supporting the safety and effectiveness of AUGMENT Bone Graft and leverages clinical data from the U.S. and Canada for the injectable formulation.”
The U.S. launch of AUGMENT Injectable is expected to deliver sustained growth in the U.S. for the AUGMENT Regenerative Solutions platform over the next several years with a gradual ramp as the injectable form is reviewed by hospital value analysis committees and physician education programs are executed. The company will immediately start the process of finalizing its packaging carrying the FDA approved labeling and expects to begin selling product in the U.S. shortly and complete the rollout in the next four to six weeks.
Dr. Timothy Daniels, Chief of the Division of Orthopaedic Surgery of St. Michael’s Hospital and Professor at the University of Toronto in the Department of Surgery, commented, “In Canada, my colleagues and I have been using AUGMENT Injectable since 2011 based upon its ideal handling characteristics and impressive clinical results. FDA approval of AUGMENT Injectable provides a valuable enhancement to the existing AUGMENT product line for U.S. foot and ankle surgeons as a front-line biologic alternative that drives the biological healing process and eliminates the costs and clinical impacts of autograft harvesting.”
Dr. Jeremy McCormick, Associate Professor of Orthopaedic Surgery at Washington University School of Medicine in St. Louis, Mo., added, “I adopted AUGMENT Bone Graft shortly after it launched as a clinically proven tool that has improved patient outcomes. As my confidence in the product has grown, so too has my use of AUGMENT. Now, with the introduction of the new injectable version, delivery of the product to the fusion site will be simplified and more consistent, helping foot and ankle specialists use it more dependably in patients.”
AUGMENT Injectable is the first clinically proven injectable protein therapeutic to come to the U.S. orthopaedic market offering an alternative to autograft in ankle and/or hindfoot fusion procedures, which translates into an estimated market opportunity of approximately $300 million in the U.S. As FDA-approved alternatives to autograft in ankle and/or hindfoot fusion procedures, AUGMENT Bone Graft and AUGMENT Injectable offer a clear patient benefit by avoiding secondary surgical sites for the harvest of autograft tissue, which can result in prolonged harvest site pain and other comorbidities in some patients.
As a result of this approval, the company plans to update its 2018 annual guidance on its second quarter earnings call scheduled for August 8, 2018.