EINPresswire09.30.20
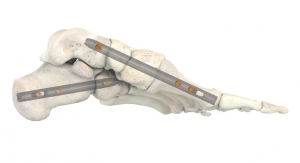
Carbon22, a GLW Inc. company, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its new FusionFrame Ring Lock System, a circular external fixator. This system was designed to be simple-to-use with a wide range of assembly components. FusionFrame was developed in part in collaboration with the Carbon22 Consortium, a select group of orthopedic and podiatric surgeons.
The design ensures circumferential access for precise fixation and can address a variety of indications in a straightforward and cost-effective manner. Multi-level constructs may be built to target the zone of pathology or apex of a deformity.
Axel Cremer, chief technology officer for Carbon22, stated, “The 510(k) clearance for our FusionFrame Ring Lock System is an exciting milestone for Carbon22. Our team has done an excellent job of expanding our circular external fixator technology platform and building on the clinical success of the existing Ring Lock System. We’re excited to launch the FusionFrame Ring Lock System and look forward to introducing future variations which are currently in development.”
The FusionFrame Ring Lock System represents next-level functionality, utilizes state-of-the-art materials, and boasts an unparalleled design that provides the ability to customize frames specific to patient needs. This system can treat complex foot and ankle deformities, as well as provide a static frame for various indications.
“As we continue to develop our new, patent-pending technology platforms, designed to allow for visualization of fusion sites (intraoperatively) and post-operative healing zones, we are working to ensure that foot and ankle surgeons have the right devices they needto treat today’s foot and ankle conditions,“ said Thomas H. Lee M.D., chief medical officer for Carbon22.
The design ensures circumferential access for precise fixation and can address a variety of indications in a straightforward and cost-effective manner. Multi-level constructs may be built to target the zone of pathology or apex of a deformity.
Axel Cremer, chief technology officer for Carbon22, stated, “The 510(k) clearance for our FusionFrame Ring Lock System is an exciting milestone for Carbon22. Our team has done an excellent job of expanding our circular external fixator technology platform and building on the clinical success of the existing Ring Lock System. We’re excited to launch the FusionFrame Ring Lock System and look forward to introducing future variations which are currently in development.”
The FusionFrame Ring Lock System represents next-level functionality, utilizes state-of-the-art materials, and boasts an unparalleled design that provides the ability to customize frames specific to patient needs. This system can treat complex foot and ankle deformities, as well as provide a static frame for various indications.
“As we continue to develop our new, patent-pending technology platforms, designed to allow for visualization of fusion sites (intraoperatively) and post-operative healing zones, we are working to ensure that foot and ankle surgeons have the right devices they needto treat today’s foot and ankle conditions,“ said Thomas H. Lee M.D., chief medical officer for Carbon22.