Sam Brusco, Associate Editor04.02.19
Last May, the U.S. Food and Drug Administration (FDA) issued an “innovation challenge”—officially titled “FDA Innovation Challenge: Devices to Prevent and Treat Opioid Use Disorder,”—to medical device companies. The FDA’s call to action intended to spur the development of medical devices, diagnostic tests, and digital health technologies (mobile health applications included) to aid in the fight against the opioid crisis and achieve the prevention and treatment of opioid use disorder (OUD).
The agency received an overwhelming response to its challenge from the medtech community. Over 250 applications were received from medical device companies of all sizes. The FDA chose eight of these participants to move forward with developing their technologies under the program. Similar to the agency’s Breakthrough Devices Program, each applicant will work with the FDA to accelerate development and expedite marketing application. Upon completion of the challenge, each device will be awarded Breakthrough Device designation.
Avanos Medical, an Alpharetta, Ga.-based company focused on creating the next generation of healthcare solutions that address reducing the use of opioids while helping patients move from surgery to recovery, is one of the eights companies taking on the FDA’s challenge. Avanos is currently working closely with the FDA to commercialize its enhanced nerve visualization system to help eliminate or reduce surgical patients’ opioid use. The company’s portfolio already consists of a number of pain control technologies—the ON-Q* Pain Relief System, an opioid-sparing elastomeric ambulatory infusion pump; COOLIEF* Cooled Radiofrequency for minimally invasive, non-opioid chronic pain treatment; and the recently acquired Game Ready cold and intermittent compression therapy system for injury and surgical recovery.
To gain more insight into how technology can help to curb or eliminate opioid use, I recently spoke with John Tushar, Avanos Medical’s president of global franchises. Some of his input is included in the pain management-focused cover story for the March/April issue of ODT, entitled “Pain, Pain, Go Away.” The full extent of John’s comments are featured here.
Sam Brusco: Why is it important for medical technology to be a part of a pain management regimen?
John Tushar: There is no one-size-fits-all approach to pain management and each person experiences pain differently. While opioid prescriptions have long been a viable treatment for a variety of pain conditions, they have led to the drug addiction epidemic and new ways to successfully and safely treat pain are necessary for today’s patients. Innovative solutions to pain management such as device alternatives can help patients optimize their pain control to best meet their needs and improve care, while simultaneously reducing the need for opioids.
COOLIEF* Cooled RF is the only currently known thermal radiofrequency system using water-cooled technology to deactivate pain-causing sensory nerves.
Brusco: What solutions does Avanos offer for acute and chronic pain, and how do they work?
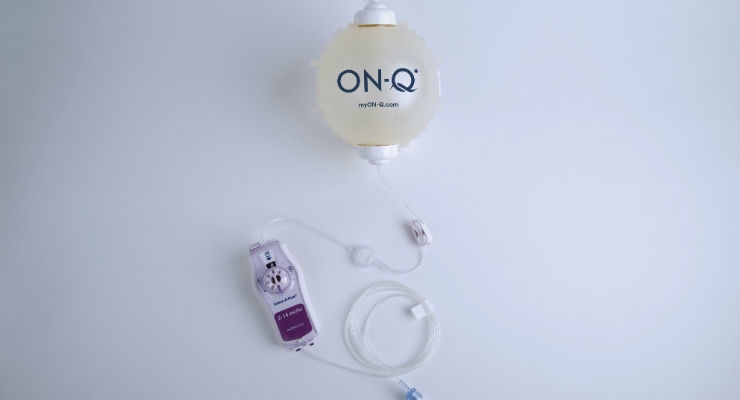
Tushar: We currently offer therapies in non-opioid acute and chronic pain management. The ON-Q* Pain Relief System is an opioid-sparing elastomeric ambulatory infusion pump system indicated to significantly reduce the need for opioids and provide better pain relief than opioids alone. It delivers a customizable and continuous flow of local anesthetic to a patient's surgical site or in close proximity to peripheral nerves and can be used for up to five days after surgery. Patients treated with ON-Q* may experience faster recovery and less post-operative pain with fewer side effects than patients taking opioids alone. ON-Q* may help surgical patients to avoid taking that first opioid, which could otherwise lead to an addiction.
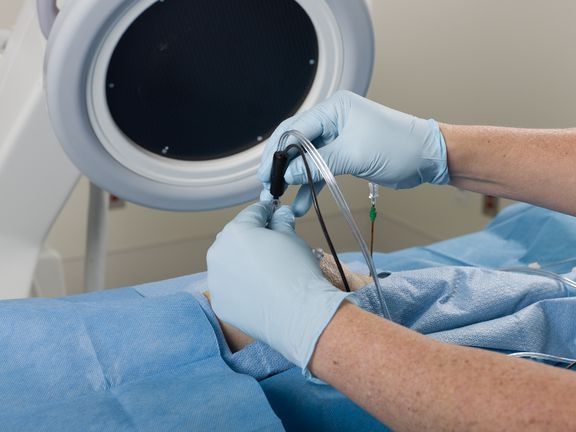
COOLIEF* Cooled Radiofrequency (RF) is a non-opioid, minimally invasive, non-surgical outpatient procedure providing long-lasting relief for chronic pain patients. Although prescription medications are commonly given to treat chronic pain, they do not effectively target the specific nerves transmitting pain and may come with a variety of negative side effects. COOLIEF* Cooled RF is proven by level I evidence in multiple modalities and works by using water-cooled technology to safely deactivate nerves sending pain signals to the brain.
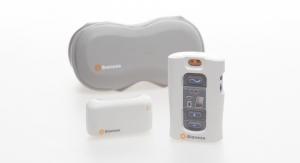
Last year, we acquired Game Ready, a clinically-proven cold and intermittent compression therapy system used for injury and orthopedic surgery recovery. The Game Ready System delivers targeted cold and compression therapy through anatomically-designed wraps to help to reduce pain, swelling, and opioid consumption. Adding this market-leading technology to the Avanos portfolio of non-opioid treatment options further commits the company to the innovative fight to reduce the use of opioids.
Brusco: Can you comment on Avanos’ participation in the FDA’s Opioid Addiction Innovation Challenge?
Tushar: In late 2018, we were selected as one of eight companies worldwide to participate in the innovation challenge to tackle the opioid epidemic. As a winner, we are working closely with the FDA to get our enhanced nerve visualization system cleared for commercialization. Our selection further supports the company’s initiative to provide innovative non-opioid therapies to eliminate or reduce the use of opioids for surgical patients.

The Game Ready GRPro 2.1 system features ACCEL Technology (Active Compression and Cold Exchange Loop), enabling the integrated delivery of continuous cold and intermittent pneumatic compression therapies.
Brusco: Are you finding that clinicians, patients, and payors are embracing medical technology as a solution to manage pain? If so, what do you believe convinced them to use the technology over traditional pain management methods?
Tushar: Yes, I think so, but there is still room for improvement. In response to the opioid crisis, healthcare providers and their patients are increasingly turning to opioid reduction strategies through proven approaches and existing device alternatives, such as ON-Q*, for opioid-sparing pain relief. Physician and patient education has led to a reduction in the number of opioids patients take after surgery and helped to improve patient outcomes. The evidence goes a long way in encouraging providers to embrace medical technology. Congress also recently passed the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act, which includes provisions related to Medicare reimbursement and requires evaluation of whether the current Medicare payment structure incentivizes opioid use over non-opioid alternatives. These efforts can also impact the adoption of non-opioid pain management solutions.
The agency received an overwhelming response to its challenge from the medtech community. Over 250 applications were received from medical device companies of all sizes. The FDA chose eight of these participants to move forward with developing their technologies under the program. Similar to the agency’s Breakthrough Devices Program, each applicant will work with the FDA to accelerate development and expedite marketing application. Upon completion of the challenge, each device will be awarded Breakthrough Device designation.
Avanos Medical, an Alpharetta, Ga.-based company focused on creating the next generation of healthcare solutions that address reducing the use of opioids while helping patients move from surgery to recovery, is one of the eights companies taking on the FDA’s challenge. Avanos is currently working closely with the FDA to commercialize its enhanced nerve visualization system to help eliminate or reduce surgical patients’ opioid use. The company’s portfolio already consists of a number of pain control technologies—the ON-Q* Pain Relief System, an opioid-sparing elastomeric ambulatory infusion pump; COOLIEF* Cooled Radiofrequency for minimally invasive, non-opioid chronic pain treatment; and the recently acquired Game Ready cold and intermittent compression therapy system for injury and surgical recovery.
To gain more insight into how technology can help to curb or eliminate opioid use, I recently spoke with John Tushar, Avanos Medical’s president of global franchises. Some of his input is included in the pain management-focused cover story for the March/April issue of ODT, entitled “Pain, Pain, Go Away.” The full extent of John’s comments are featured here.
Sam Brusco: Why is it important for medical technology to be a part of a pain management regimen?
John Tushar: There is no one-size-fits-all approach to pain management and each person experiences pain differently. While opioid prescriptions have long been a viable treatment for a variety of pain conditions, they have led to the drug addiction epidemic and new ways to successfully and safely treat pain are necessary for today’s patients. Innovative solutions to pain management such as device alternatives can help patients optimize their pain control to best meet their needs and improve care, while simultaneously reducing the need for opioids.

COOLIEF* Cooled RF is the only currently known thermal radiofrequency system using water-cooled technology to deactivate pain-causing sensory nerves.
Brusco: What solutions does Avanos offer for acute and chronic pain, and how do they work?
Tushar: We currently offer therapies in non-opioid acute and chronic pain management. The ON-Q* Pain Relief System is an opioid-sparing elastomeric ambulatory infusion pump system indicated to significantly reduce the need for opioids and provide better pain relief than opioids alone. It delivers a customizable and continuous flow of local anesthetic to a patient's surgical site or in close proximity to peripheral nerves and can be used for up to five days after surgery. Patients treated with ON-Q* may experience faster recovery and less post-operative pain with fewer side effects than patients taking opioids alone. ON-Q* may help surgical patients to avoid taking that first opioid, which could otherwise lead to an addiction.
COOLIEF* Cooled Radiofrequency (RF) is a non-opioid, minimally invasive, non-surgical outpatient procedure providing long-lasting relief for chronic pain patients. Although prescription medications are commonly given to treat chronic pain, they do not effectively target the specific nerves transmitting pain and may come with a variety of negative side effects. COOLIEF* Cooled RF is proven by level I evidence in multiple modalities and works by using water-cooled technology to safely deactivate nerves sending pain signals to the brain.
Last year, we acquired Game Ready, a clinically-proven cold and intermittent compression therapy system used for injury and orthopedic surgery recovery. The Game Ready System delivers targeted cold and compression therapy through anatomically-designed wraps to help to reduce pain, swelling, and opioid consumption. Adding this market-leading technology to the Avanos portfolio of non-opioid treatment options further commits the company to the innovative fight to reduce the use of opioids.
Brusco: Can you comment on Avanos’ participation in the FDA’s Opioid Addiction Innovation Challenge?
Tushar: In late 2018, we were selected as one of eight companies worldwide to participate in the innovation challenge to tackle the opioid epidemic. As a winner, we are working closely with the FDA to get our enhanced nerve visualization system cleared for commercialization. Our selection further supports the company’s initiative to provide innovative non-opioid therapies to eliminate or reduce the use of opioids for surgical patients.

The Game Ready GRPro 2.1 system features ACCEL Technology (Active Compression and Cold Exchange Loop), enabling the integrated delivery of continuous cold and intermittent pneumatic compression therapies.
Brusco: Are you finding that clinicians, patients, and payors are embracing medical technology as a solution to manage pain? If so, what do you believe convinced them to use the technology over traditional pain management methods?
Tushar: Yes, I think so, but there is still room for improvement. In response to the opioid crisis, healthcare providers and their patients are increasingly turning to opioid reduction strategies through proven approaches and existing device alternatives, such as ON-Q*, for opioid-sparing pain relief. Physician and patient education has led to a reduction in the number of opioids patients take after surgery and helped to improve patient outcomes. The evidence goes a long way in encouraging providers to embrace medical technology. Congress also recently passed the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act, which includes provisions related to Medicare reimbursement and requires evaluation of whether the current Medicare payment structure incentivizes opioid use over non-opioid alternatives. These efforts can also impact the adoption of non-opioid pain management solutions.