08.15.17
$11.3 Billion
KEY EXECUTIVES:
Kevin A. Lobo, Chairman and CEO
Glenn S. Boehnlein, VP, CFO
Lonny J. Carpenter, Group President, Global Quality and Business Operations
David K. Floyd, Group President, Orthopaedics
Timothy J. Scannell, Group President, MedSurg & Neurotechnology
Xavier M. C. Berling, President, Trauma & Extremities
William J. Huffnagle, President, Joint Replacement
Bradley W. Paddock, President, Spine
Mark H. Paul, President, Neurovascular
J. Andrew Pierce, President, Endoscopy
Bradford L. Saar, President, Medical
Spencer S. Stiles, President, Instruments
Brian J. White, President, Sustainability Solutions
NUMBER OF EMPLOYEES: 33,000
GLOBAL HEADQUARTERS: Kalamazoo, Mich.
Mergers and acquisitions are a large part of the OEM environment in today’s medical device space. Companies are seeking strategic targets to purchase as a rapid method with which to enhance their international footprint, improve product offerings in specific device sectors, or supplement R&D with the procurement of a startup company that’s developing a cutting-edge technology. It’s become a pervasive element in the strategy of most medtech OEMs. Stryker, however, may have been one of the more active in 2016. Perhaps not in total dollars, but in terms of the number of transactions, they were likely near the top of the list. And looking at the company’s revenues for the 2016 fiscal year, it seems to be a profitable success.
Overall, Stryker invested more than $4 billion into M&A activities in fiscal 2016. Its two larger purchases were Sage Products and Physio-Control International. In addition, it bought the SafeWire product portfolio, Synergetics USA’s neuro portfolio, Stanmore Implants Worldwide, Ivy Sports Medicine, and BD’s vertebral compression fracture portfolio.
On Feb. 1, Stryker announced it was purchasing Sage Products from Madison Dearborn Partners at a price tag of $2.775 billion. The firm produces disposable products intended to reduce “never events” within the intensive care unit and MedSurg hospital unit setting. The company’s products address issues such as hospital-acquired infections (HAIs), skin injuries, and healthcare worker injury. Sales of these products in 2015 totaled $430 million, which was an increase of 13 percent over the prior year. Undoubtedly, with the shift to value-based care and the efforts toward reducing the instances of such events (particularly HAIs), Stryker views the company as a growth opportunity.
“The company’s established leadership team and innovative products that help prevent hospital-acquired conditions have driven consistent double-digit sales growth,” Kevin A. Lobo, chairman and CEO of Stryker, stated in a release first announcing the transaction. “This acquisition aligns with Stryker’s focus on offering products and services that support a mindset of prevention, specifically in the area of ‘never events’ such as hospital-acquired infections. Today, through our Medical division, Stryker offers products that are complementary to those produced by Sage. Sage has a 45-year history of focus on patients and caregivers that is evident in their culture and fits well with our Medical division. This business will also provide a consistent disposable revenue stream that will complement our capital equipment offerings. We look forward to welcoming the Sage team to Stryker.”
The deal formally closed only a couple of months later, on April 1, 2016. Four days later, Stryker noted the completion of its other significant deal in fiscal 2016—the acquisition of Physio-Control International Inc. This transaction—valued at $1.28 billion—was first announced on Feb. 16, 2016. The firm was sold to Stryker by Bain Capital Private Equity in an all-cash transaction. Physio-Control offers more of an emergency medicine type of product line to Stryker, with monitors/defibrillators, automated external defibrillators, and CPR-assist devices making up the bulk of the company’s offerings. It also offers data management and support services. Sales in 2015 totaled $503 million, which was an increase over the prior year by 6 percent.
Another valuable aspect of Physio-Control was its established presence and infrastructure internationally. “Physio-Control has achieved global leadership positions with a strong brand and customer-centered solutions that can predict or intervene in life-threatening emergencies,” Lobo stated in a release first announcing the transaction. “Physio-Control’s focused strategy and their culture will fit well within the EMS business of our Medical division, further leveraging our existing call pattern. We look forward to welcoming the Physio-Control team to Stryker.”
While these two were the larger transactions for Stryker in 2016, the other deals the OEM made were still significant. Starting in February, it was announced that the neuro portfolio of Synergetics USA was to shift to Stryker in an all-cash deal. This portfolio is comprised of products including the Malis generator, Spetzler Malis disposable forceps, and Stryker’s existing Sonopet tips and RF generator. Sales of approximately $31 million were attributed to the portfolio in 2015.
“The acquisition of the Synergetics neuro portfolio is highly complementary to Stryker Instruments’ Neuro Spine & ENT (NSE) business and is aligned with NSE’s strategy of expanding its neurosurgical product offering,” Timothy J. Scannell, group president of MedSurg and Neurotechnology, stated at the time of the announcement.
Shortly after the Synergetics deal was completed (April 1, 2016), Stryker had another M&A announcement. On April 10, the OEM declared that it was purchasing the SafeWire product portfolio, which had been composed of the Y-Wire guidewire and the Tiger Jamshidi Needle family for use in minimally invasive spine surgery. The products were purchased to enhance Stryker’s spine product portfolio and its focus on minimally invasive spine surgery offerings.
In a release issued announcing the acquisition, Brad Paddock, president of Stryker’s Spine division, said the introduction of the SafeWire portfolio reflects Stryker’s ongoing commitment to patient safety and providing surgeons with innovative technology needed to treat patients undergoing minimally invasive spine surgery. “This acquisition increases our competitive advantage as we broaden our product line and extend our customer base among teaching facilities, competitive accounts, and existing SafeWire customers.”
Another April acquisition was the purchase of BD’s vertebral augmentation solutions business, which included the AVAmax, AVAtex, and AVAprep brands. These minimally invasive products are used in vertebroplasty and vertebral augmentation procedures. They originally came out of the BD acquisition of CareFusion. Similar to several aforementioned transactions, this purchase was accomplished with the intent of bolstering Stryker’s preexisting product line in the space.
“The acquisition of BD’s VCF portfolio is highly complementary to Stryker Instruments’ Interventional Spine (IVS) business and is aligned with IVS’s strategy of expanding its VCF footprint,” Scannell stated at the time of the deal announcement. “It also further strengthens Stryker’s brand in the Neurotechnology space.”
ANALYST INSIGHTS: Despite solid performance in recent years, Stryker’s biggest challenge will be to successfully sell outside North America. The company sees approximately 70 percent of revenues from the U.S. and Canada while these markets now represent under 50 percent of the total market.
The purchase of Stanmore Implants Worldwide Limited from SIW Holdings Limited for a purchase price of £35.6 million rounded out Stryker’s April buying spree. Stanmore offers custom and off-the-shelf solutions that address the needs of surgeons treating adult and juvenile orthopedic oncology.
Although quiet through the summer months, Stryker kicked off September with its final M&A declaration in fiscal 2016 with the announcement of the acquisition of Ivy Sports Medicine LLC. Ivy Sports Medicine’s product offering was centered around the minimally invasive repair of the meniscus. The portfolio included the only FDA-approved collagen meniscus implant (CMI) and an inside-out meniscal suturing platform.
“The acquisition of Ivy Sports Medicine strengthens our capabilities and fits strategically with our current portfolio. Ivy’s complete meniscal platform, coupled with their clinical history, will allow us to provide our customers with multiple solutions to address meniscal repair,” Matt Moreau, vice president and general manager of Stryker’s Sports Medicine business, said at the time of the announcement. “This is an area of sports medicine where there is continued opportunity to address unmet customer needs. The Ivy portfolio provides a unique platform for us to build upon as we seek to continue advancing the treatment of meniscal injuries.”
Undoubtedly as a result of the completion of several of these transactions within fiscal 2016, coupled with the success of acquisitions made in earlier years as well as organic sales growth of Stryker’s legacy and newly released products, the OEM enjoyed a significant boost in sales in 2016. For the first time in its history, the company bypassed both the $10 billion and $11 billion sales marks to finish the year at $11.3 billion (an increase of 13.9 percent over the prior year), compared to $9.9 billion in 2015 and $9.7 billion in 2014. Possibly a concern for future growth (or a great opportunity, depending upon how it’s viewed), the company’s sales in fiscal 2016 were primarily driven by the United States, which accounted for 73 percent of total revenue. Across the board, the company’s three main segments—Orthopaedics, MedSurg, and Neurotechnology and Spine—will need to better penetrate international markets to enjoy even greater growth in the years to come.
The Orthopaedics segment is primarily focused on implants for knee and hip joint replacements, as well as trauma and extremity surgeries. Notable within the segment is the company’s Mako total knee application, which joined the company’s other Mako offerings in the robotic arm-assisted reconstructive surgery line after it received FDA clearance in 2015. The company celebrated the full launch of the total knee in March 2017. Looking at the segment’s full offering, sales break down fairly evenly among several divisions. Knees were responsible for $1.5 billion in sales, followed by Trauma and Extremities at $1.4 billion. Hips came in at $1.3 billion, with “Other” accounting for $285 million, totaling $4.4 billion for the entire segment. This represented an increase of 4.7 percent over 2015.
MedSurg, which bypassed Orthopaedics in 2016 in terms of percentage of overall contribution to the company’s sales figure for the first time in several years (43 percent for MedSurg versus 39 percent for Orthopaedics), was bolstered by several of the aforementioned acquisitions. The end result was a substantial 25.6 percent increase in sales over 2015. The product offerings from this segment include surgical equipment and navigation systems (Instruments); endoscopic and communications systems (Endoscopy); patient handling, emergency medical equipment, and intensive care disposable products (Medical); and reprocessed and remanufactured medical devices (Sustainability). Similar to the Orthopaedics segment, the sales are spread almost evenly across the divisions, totaling $4.9 billion in sales. Medical led the segment’s sales with more than $1.6 billion, followed by Instruments at just under $1.6 billion. Endoscopy was close behind with $1.5 billion and at a distant fourth for the segment was Sustainability at $238 million.
Neurotechnology and Spine (the name also reflects the only two divisions within the segment) contributed 18 percent to the company’s total sales. The Neurotechnology offering includes products used for minimally invasive endovascular techniques; a line of products for traditional brain and open skull-based surgical procedures; orthobiologic and biosurgery products, including synthetic bone grafts and vertebral augmentation products; and minimally invasive products for the treatment of acute ischemic and hemorrhagic stroke. The Spine products include cervical, thoracolumbar, and interbody systems used in spinal injury, deformity, and degenerative therapies. Sales translated to a 62/38 percent split with Neuro contributing almost $1.3 billion to sales, while Spine was at $754 million. In total, the segment’s $2 billion in sales reflected a 9.9 percent increase over the prior year.
Outside of the product and company acquisitions, Stryker reported organic growth of approximately 6 percent over 2015. While much of that was from already established products the company provided, some of the growth may have originated from several of the new offerings announced in fiscal 2016. In March, the OEM formally introduced two new products at the American Academy of Orthopaedic Surgeons annual meeting—the Aero-C Cervical Stability System and the Xia 4.5 Cortical Trajectory Implants and Instruments.
ANALYST INSIGHTS: Never a shy company, watch for Stryker to continue its aggressive growth through both acquisition and its international expansion efforts. Stryker is a dangerous competitor as it wakes up “unafraid” to challenge itself each and every day!
The company claims the Spine division’s Aero-C is the only straight-forward ACDF device that offers uniform compression across the interbody space. The technology received FDA 510(k) clearance in December 2015. Also out of the Spine division, the Xia products are used in less invasive LITe LIF posterior lumbar interbody fusion procedures for patients with degenerative disc disease, spondylolisthesis, and trauma. According to the company, it is also intended to be more muscle-sparing than standard open procedures that require lateral dissection.
Keeping busy with product launches, the Spine division also debuted a 3D-printed Tritanium posterior lumbar cage at the 2016 American Association of Neurological Surgeons annual meeting. According to Stryker, Tritanium is a novel, highly porous titanium alloy material designed for bone in-growth and biological fixation.
Perhaps addressing the potential challenge associated with the company’s international sales figures, in November 2016, Stryker announced a 20-year partnership with Indo UK Institute of Health’s (IUIH) Medicity Program. Under terms of the agreement, the OEM would serve as a “preferred partner” in the orthopedic areas of hip, knee, and trauma products and services. The deal also includes a sponsorship by Stryker of a post-graduate training education center on the campus of one of IUIH’s medicities and R&D collaboration with Stryker’s Global Technology Center in India.
“The partnership with Stryker is set to redefine the existing paradigms in [the] healthcare industry,” said Ajay Rajan Gupta, managing director and group CEO, IUIH. “With predictable orthopedic spend, improved cash flow, and optimized use of technology resources, the agreement with Stryker will enhance IUIH’s value in healthcare space in years to come.”
KEY EXECUTIVES:
Kevin A. Lobo, Chairman and CEO
Glenn S. Boehnlein, VP, CFO
Lonny J. Carpenter, Group President, Global Quality and Business Operations
David K. Floyd, Group President, Orthopaedics
Timothy J. Scannell, Group President, MedSurg & Neurotechnology
Xavier M. C. Berling, President, Trauma & Extremities
William J. Huffnagle, President, Joint Replacement
Bradley W. Paddock, President, Spine
Mark H. Paul, President, Neurovascular
J. Andrew Pierce, President, Endoscopy
Bradford L. Saar, President, Medical
Spencer S. Stiles, President, Instruments
Brian J. White, President, Sustainability Solutions
NUMBER OF EMPLOYEES: 33,000
GLOBAL HEADQUARTERS: Kalamazoo, Mich.
Mergers and acquisitions are a large part of the OEM environment in today’s medical device space. Companies are seeking strategic targets to purchase as a rapid method with which to enhance their international footprint, improve product offerings in specific device sectors, or supplement R&D with the procurement of a startup company that’s developing a cutting-edge technology. It’s become a pervasive element in the strategy of most medtech OEMs. Stryker, however, may have been one of the more active in 2016. Perhaps not in total dollars, but in terms of the number of transactions, they were likely near the top of the list. And looking at the company’s revenues for the 2016 fiscal year, it seems to be a profitable success.
Overall, Stryker invested more than $4 billion into M&A activities in fiscal 2016. Its two larger purchases were Sage Products and Physio-Control International. In addition, it bought the SafeWire product portfolio, Synergetics USA’s neuro portfolio, Stanmore Implants Worldwide, Ivy Sports Medicine, and BD’s vertebral compression fracture portfolio.
On Feb. 1, Stryker announced it was purchasing Sage Products from Madison Dearborn Partners at a price tag of $2.775 billion. The firm produces disposable products intended to reduce “never events” within the intensive care unit and MedSurg hospital unit setting. The company’s products address issues such as hospital-acquired infections (HAIs), skin injuries, and healthcare worker injury. Sales of these products in 2015 totaled $430 million, which was an increase of 13 percent over the prior year. Undoubtedly, with the shift to value-based care and the efforts toward reducing the instances of such events (particularly HAIs), Stryker views the company as a growth opportunity.
“The company’s established leadership team and innovative products that help prevent hospital-acquired conditions have driven consistent double-digit sales growth,” Kevin A. Lobo, chairman and CEO of Stryker, stated in a release first announcing the transaction. “This acquisition aligns with Stryker’s focus on offering products and services that support a mindset of prevention, specifically in the area of ‘never events’ such as hospital-acquired infections. Today, through our Medical division, Stryker offers products that are complementary to those produced by Sage. Sage has a 45-year history of focus on patients and caregivers that is evident in their culture and fits well with our Medical division. This business will also provide a consistent disposable revenue stream that will complement our capital equipment offerings. We look forward to welcoming the Sage team to Stryker.”
The deal formally closed only a couple of months later, on April 1, 2016. Four days later, Stryker noted the completion of its other significant deal in fiscal 2016—the acquisition of Physio-Control International Inc. This transaction—valued at $1.28 billion—was first announced on Feb. 16, 2016. The firm was sold to Stryker by Bain Capital Private Equity in an all-cash transaction. Physio-Control offers more of an emergency medicine type of product line to Stryker, with monitors/defibrillators, automated external defibrillators, and CPR-assist devices making up the bulk of the company’s offerings. It also offers data management and support services. Sales in 2015 totaled $503 million, which was an increase over the prior year by 6 percent.
Another valuable aspect of Physio-Control was its established presence and infrastructure internationally. “Physio-Control has achieved global leadership positions with a strong brand and customer-centered solutions that can predict or intervene in life-threatening emergencies,” Lobo stated in a release first announcing the transaction. “Physio-Control’s focused strategy and their culture will fit well within the EMS business of our Medical division, further leveraging our existing call pattern. We look forward to welcoming the Physio-Control team to Stryker.”
While these two were the larger transactions for Stryker in 2016, the other deals the OEM made were still significant. Starting in February, it was announced that the neuro portfolio of Synergetics USA was to shift to Stryker in an all-cash deal. This portfolio is comprised of products including the Malis generator, Spetzler Malis disposable forceps, and Stryker’s existing Sonopet tips and RF generator. Sales of approximately $31 million were attributed to the portfolio in 2015.
“The acquisition of the Synergetics neuro portfolio is highly complementary to Stryker Instruments’ Neuro Spine & ENT (NSE) business and is aligned with NSE’s strategy of expanding its neurosurgical product offering,” Timothy J. Scannell, group president of MedSurg and Neurotechnology, stated at the time of the announcement.
Shortly after the Synergetics deal was completed (April 1, 2016), Stryker had another M&A announcement. On April 10, the OEM declared that it was purchasing the SafeWire product portfolio, which had been composed of the Y-Wire guidewire and the Tiger Jamshidi Needle family for use in minimally invasive spine surgery. The products were purchased to enhance Stryker’s spine product portfolio and its focus on minimally invasive spine surgery offerings.
In a release issued announcing the acquisition, Brad Paddock, president of Stryker’s Spine division, said the introduction of the SafeWire portfolio reflects Stryker’s ongoing commitment to patient safety and providing surgeons with innovative technology needed to treat patients undergoing minimally invasive spine surgery. “This acquisition increases our competitive advantage as we broaden our product line and extend our customer base among teaching facilities, competitive accounts, and existing SafeWire customers.”
Another April acquisition was the purchase of BD’s vertebral augmentation solutions business, which included the AVAmax, AVAtex, and AVAprep brands. These minimally invasive products are used in vertebroplasty and vertebral augmentation procedures. They originally came out of the BD acquisition of CareFusion. Similar to several aforementioned transactions, this purchase was accomplished with the intent of bolstering Stryker’s preexisting product line in the space.
“The acquisition of BD’s VCF portfolio is highly complementary to Stryker Instruments’ Interventional Spine (IVS) business and is aligned with IVS’s strategy of expanding its VCF footprint,” Scannell stated at the time of the deal announcement. “It also further strengthens Stryker’s brand in the Neurotechnology space.”
ANALYST INSIGHTS: Despite solid performance in recent years, Stryker’s biggest challenge will be to successfully sell outside North America. The company sees approximately 70 percent of revenues from the U.S. and Canada while these markets now represent under 50 percent of the total market.
—Tony Freeman, President, AS Freeman Advisors LLC
The purchase of Stanmore Implants Worldwide Limited from SIW Holdings Limited for a purchase price of £35.6 million rounded out Stryker’s April buying spree. Stanmore offers custom and off-the-shelf solutions that address the needs of surgeons treating adult and juvenile orthopedic oncology.
Although quiet through the summer months, Stryker kicked off September with its final M&A declaration in fiscal 2016 with the announcement of the acquisition of Ivy Sports Medicine LLC. Ivy Sports Medicine’s product offering was centered around the minimally invasive repair of the meniscus. The portfolio included the only FDA-approved collagen meniscus implant (CMI) and an inside-out meniscal suturing platform.
“The acquisition of Ivy Sports Medicine strengthens our capabilities and fits strategically with our current portfolio. Ivy’s complete meniscal platform, coupled with their clinical history, will allow us to provide our customers with multiple solutions to address meniscal repair,” Matt Moreau, vice president and general manager of Stryker’s Sports Medicine business, said at the time of the announcement. “This is an area of sports medicine where there is continued opportunity to address unmet customer needs. The Ivy portfolio provides a unique platform for us to build upon as we seek to continue advancing the treatment of meniscal injuries.”
Undoubtedly as a result of the completion of several of these transactions within fiscal 2016, coupled with the success of acquisitions made in earlier years as well as organic sales growth of Stryker’s legacy and newly released products, the OEM enjoyed a significant boost in sales in 2016. For the first time in its history, the company bypassed both the $10 billion and $11 billion sales marks to finish the year at $11.3 billion (an increase of 13.9 percent over the prior year), compared to $9.9 billion in 2015 and $9.7 billion in 2014. Possibly a concern for future growth (or a great opportunity, depending upon how it’s viewed), the company’s sales in fiscal 2016 were primarily driven by the United States, which accounted for 73 percent of total revenue. Across the board, the company’s three main segments—Orthopaedics, MedSurg, and Neurotechnology and Spine—will need to better penetrate international markets to enjoy even greater growth in the years to come.
The Orthopaedics segment is primarily focused on implants for knee and hip joint replacements, as well as trauma and extremity surgeries. Notable within the segment is the company’s Mako total knee application, which joined the company’s other Mako offerings in the robotic arm-assisted reconstructive surgery line after it received FDA clearance in 2015. The company celebrated the full launch of the total knee in March 2017. Looking at the segment’s full offering, sales break down fairly evenly among several divisions. Knees were responsible for $1.5 billion in sales, followed by Trauma and Extremities at $1.4 billion. Hips came in at $1.3 billion, with “Other” accounting for $285 million, totaling $4.4 billion for the entire segment. This represented an increase of 4.7 percent over 2015.
MedSurg, which bypassed Orthopaedics in 2016 in terms of percentage of overall contribution to the company’s sales figure for the first time in several years (43 percent for MedSurg versus 39 percent for Orthopaedics), was bolstered by several of the aforementioned acquisitions. The end result was a substantial 25.6 percent increase in sales over 2015. The product offerings from this segment include surgical equipment and navigation systems (Instruments); endoscopic and communications systems (Endoscopy); patient handling, emergency medical equipment, and intensive care disposable products (Medical); and reprocessed and remanufactured medical devices (Sustainability). Similar to the Orthopaedics segment, the sales are spread almost evenly across the divisions, totaling $4.9 billion in sales. Medical led the segment’s sales with more than $1.6 billion, followed by Instruments at just under $1.6 billion. Endoscopy was close behind with $1.5 billion and at a distant fourth for the segment was Sustainability at $238 million.
Neurotechnology and Spine (the name also reflects the only two divisions within the segment) contributed 18 percent to the company’s total sales. The Neurotechnology offering includes products used for minimally invasive endovascular techniques; a line of products for traditional brain and open skull-based surgical procedures; orthobiologic and biosurgery products, including synthetic bone grafts and vertebral augmentation products; and minimally invasive products for the treatment of acute ischemic and hemorrhagic stroke. The Spine products include cervical, thoracolumbar, and interbody systems used in spinal injury, deformity, and degenerative therapies. Sales translated to a 62/38 percent split with Neuro contributing almost $1.3 billion to sales, while Spine was at $754 million. In total, the segment’s $2 billion in sales reflected a 9.9 percent increase over the prior year.
Outside of the product and company acquisitions, Stryker reported organic growth of approximately 6 percent over 2015. While much of that was from already established products the company provided, some of the growth may have originated from several of the new offerings announced in fiscal 2016. In March, the OEM formally introduced two new products at the American Academy of Orthopaedic Surgeons annual meeting—the Aero-C Cervical Stability System and the Xia 4.5 Cortical Trajectory Implants and Instruments.
ANALYST INSIGHTS: Never a shy company, watch for Stryker to continue its aggressive growth through both acquisition and its international expansion efforts. Stryker is a dangerous competitor as it wakes up “unafraid” to challenge itself each and every day!
—Dave Sheppard, Co-Founder and Principal, MedWorld Advisors
The company claims the Spine division’s Aero-C is the only straight-forward ACDF device that offers uniform compression across the interbody space. The technology received FDA 510(k) clearance in December 2015. Also out of the Spine division, the Xia products are used in less invasive LITe LIF posterior lumbar interbody fusion procedures for patients with degenerative disc disease, spondylolisthesis, and trauma. According to the company, it is also intended to be more muscle-sparing than standard open procedures that require lateral dissection.
Keeping busy with product launches, the Spine division also debuted a 3D-printed Tritanium posterior lumbar cage at the 2016 American Association of Neurological Surgeons annual meeting. According to Stryker, Tritanium is a novel, highly porous titanium alloy material designed for bone in-growth and biological fixation.
Perhaps addressing the potential challenge associated with the company’s international sales figures, in November 2016, Stryker announced a 20-year partnership with Indo UK Institute of Health’s (IUIH) Medicity Program. Under terms of the agreement, the OEM would serve as a “preferred partner” in the orthopedic areas of hip, knee, and trauma products and services. The deal also includes a sponsorship by Stryker of a post-graduate training education center on the campus of one of IUIH’s medicities and R&D collaboration with Stryker’s Global Technology Center in India.
“The partnership with Stryker is set to redefine the existing paradigms in [the] healthcare industry,” said Ajay Rajan Gupta, managing director and group CEO, IUIH. “With predictable orthopedic spend, improved cash flow, and optimized use of technology resources, the agreement with Stryker will enhance IUIH’s value in healthcare space in years to come.”
|
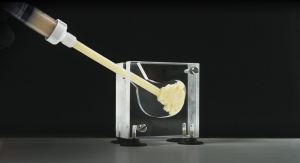
Bradley Paddock • President, Stryker Spine division A variety of bone graft materials are used in spinal fusion surgery to help promote fusion and stimulate bone healing. The graft is placed in the space between the vertebrae to be fused, often within a spinal implant or “cage,” to increase bone production and help the vertebrae heal together. Bone graft may be autologous (bone harvested from the patient’s body), allograft (obtained from a cadaveric bone bank), synthetic,* or other material, depending on surgeon needs and preferences.1 The need to deliver bone graft to the surgical site in a controlled and consistent manner is crucial, but traditional bone graft delivery methods may present challenges. These include impaction or force of funnel and mallet, issues with visualization (especially during less-invasive procedures), the inability to consistently control the precise amount of biologic material delivered to the site, and the jamming of bone funnels, which can cost surgeons time and increase frustration. These challenges present opportunities for innovation, as reflected in the development of a new hand-held device designed to facilitate delivery of bone graft material to spinal surgery sites. The LITe BIO Delivery System from Stryker Spine division simplifies graft delivery, accommodates a surgeon’s preferred graft materials,2 and allows for direct visualization of graft placement—a unique combination of features that no other bone graft delivery system provides. The system can be used for any type of spine fusion surgery, including minimally invasive procedures. Advanced Delivery, Simplified The distinct design of the BIO Delivery System, which received U.S. Food and Drug Administration clearance in 2016,2 was inspired by a caulking gun. Surgeons, nurses, surgical techs, and others provided input and feedback during development, and rigorous testing was performed to ensure the device met clinical requirements as well as Stryker’s quality standards. The sleek design provides surgeons with a single-handed method to deliver any type of autograft, allograft, or synthetic bone graft material without obstructing visibility, and the mallet-free system eliminates the impaction of bone graft.2 Materials for each component of the device were carefully selected, taking into account testing requirements and standards, the financial cost to surgeons/hospitals, and green initiatives limiting waste. The reusable handle is made of stainless steel—sturdy and strong in the surgeon’s hand and under compression of the spine. The sterile-packaged cannulas, which allow for delivery of up to 5cc of bone graft, are made of a disposable plastic material with a polycarbonate coating on the inside designed to prevent jamming and allow the device to deliver a variety of biologic materials. The delivery tool provides surgeons with tactile, audible, and visual confirmation of bone graft delivery. Its familiar tactile design is comparable to the ratcheting handle used for general surgical instruments and other spine-specific instruments. As the graft material is dispensed, the handle clicks, providing audible confirmation that the bone graft is being delivered. The low-profile design allows visibility through a decompression tube without obstructing view. This direct visualization aids in the precise placement of the graft, which is key for optimal fusion. A radiolucent strip facilitates visualization under fluoroscopy. Consistent Delivery of Bone Graft Although some conventional bone graft delivery methods can accommodate any type of bone graft, they can be cumbersome to use. Pre-filled delivery devices are more streamlined, but can only be used with specific graft materials. In contrast, the LITe BIO Delivery System was designed to accommodate any bone graft material. In addition, as adoption of the system increases, feedback indicates that surgeons appreciate the ease of use and the ability to safely deliver bone graft. “I needed a controlled mechanism for introducing bone graft into the disc space without applying a lot of force, and also without having multiple passes of bone graft material immediately adjacent to the nerve root,” said Dr. Michael J. Lee, spine surgeon and associate professor of orthopaedic surgery at The University of Chicago Medicine, and a Stryker Spine consultant.** “Stryker’s BIO Delivery System allows me to do just that.” The LITe BIO Delivery System is the latest addition to Stryker Spine division’s orthobiologics portfolio, which includes allograft chips, synthetics, demineralized bone matrix (DBM), and stem cell biologics. The one-of-a-kind bone graft delivery tool offers advanced delivery, simplified. References
** Dr. Michael J. Lee is a paid consultant of Stryker. His statements represent his own opinion based on personal experience and are not necessarily those of Stryker. Individual results may vary. Bradley Paddock has served as president of Stryker’s Spine division, based in Allendale, N.J., since 2014. He has nearly 20 years of leadership experience in medical device global commercialization. |