Search Results for 'testing'
Buyers Guide Companies
View All Buyers Guide Companies »
-
Biocompatibility & Testing
Changes Involving Biocompatibility Testing for Medical Devices—An Orthopedic Innovators Q&A
Shifting requirements from both the FDA and in the EU have generated significant questions around the laboratory evaluation of devices.
-
Biocompatibility & Testing

Updates to Conducting Toxicological Risk Assessment of Medical Device Constituents
A description of new terms and their use in the toxicological risk assessment paradigm of medical device constituents.Sharlee L. More, Ph.D., DABT, Associate Director, Toxicology, Regulatory Affairs, MCRA LLC 11.13.23
-
Biocompatibility & Testing
The Transition to MDR and Important Considerations—An Orthopedic Innovators Q&A
With the delays seemingly settled, orthopedic device manufacturers need to ensure they are aligned to the EU’s MDR.
-
Biocompatibility & Testing
Patient-Specific Implants and Proper Testing of Them—An Orthopedic Innovators Q&A
Customizing implants to a specific patient can provide fit and function advantages, but can present uncertainty with regard to testing.
-
Biocompatibility & Testing

Leveraging Automation to Improve Inspection Quality—An Orthopedic Innovators Q&A
Automation not only addresses labor shortage concerns, but also enhances the overall output by improving the inspection process.
-
Biocompatibility & Testing

Lab Tested, Industry Approved: A Discussion of Ortho Device Testing & Analysis
A group of medical/orthopedic device testing experts discuss the impacts of industry trends on their businesses.Sam Brusco, Associate Editor 09.12.23
-
Biocompatibility & Testing | Design

Exploring Issues Surrounding Quality Control & Device Design for Osteoporosis Testing & Monitoring
What are the potential solutions and how can manufacturers and orthopedic professionals provide patients with the best outcomes?Online Exclusives Amanda Winstead, Freelance Writer 08.30.23
-
Assembly & Automation | Biocompatibility & Testing

Leveraging Automated Inspection to Counter Labor Shortages
We can no longer assume competitive pay alone will be enough to attract and retain quality employees.Brad Traczyk, Orthopedic Quality Manager, Lowell Inc. 08.04.23
-
Biocompatibility & Testing | Spine/Neurology

Lifespans Launches Alfonso Cloud
The SaaS simulation platform dramatically reduces the time and cost of the implant testing process, setting a new standard for bringing new implants to market.Charles Sternberg, Associate Editor 05.15.23
-
Biocompatibility & Testing

The MDR Delay’s Impact on Regulatory and Testing—An Orthopedic Innovators Q&A
While the recent MDR delay gives companies some breathing room, it should not stall their effort in ensuring their products remain compliant for the EU.
-
Biocompatibility & Testing

Exact Metrology Successfully Scans Titanium Cervical Spine Locking Plate
The METROTOM 6 scout digitizes complex parts including the internal geometries at the finest level of detail.Charles Sternberg, Associate Editor 01.16.23
-
Biocompatibility & Testing
An Evaluation of Laboratory Testing for Orthopedic Device Makers—An Orthopedic Innovators Q&A
As devices become more complex and adopt new materials and technologies, manufacturers need laboratory testing partners who can keep pace.
-
Biocompatibility & Testing
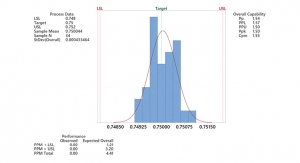
How Statistics Can Reduce Medical Device Inspection Time
Highlighting three statistical methods that can help CMs and OEMs work together to reduce inspection time.Jack Wieland , Project Manager, Lowell Inc. 09.16.22
-
Biocompatibility & Testing

Burden of Proof: An Orthopedic Testing & Analysis Roundtable
Seven orthopedic device testing and analysis experts discuss market forces, trends, and challenges in the industry.Sam Brusco, Associate Editor 09.16.22
-
Biocompatibility & Testing
The Problem Plaguing U.S. Antimicrobial Joint Implant Development
The regulatory landscape surrounding antimicrobial-associated implants is challenging to navigate.Dr. Mehdi Kazemzadeh-Narbat, Dr. Sherry Parker and Dr. Kim Ehman, WuXi AppTec 05.26.22
-
3-D Printing/Additive Mfg.

Testing and Regulatory Landscape for Additively Manufactured Devices—An Orthopedic Innovators Q&A
Uncertainty in proper procedures has left orthopedic device manufacturers seeking answers.
-
Biocompatibility & Testing
Addressing Chemical Characterization for Medical Devices—An Orthopedic Innovators Q&A
Understanding the make-up of medical devices that have direct contact with the patient is critical to ensure a safe device.
-

Could 3D Printing Simplify Orthopedic Device Testing?
Additive manufacturing has the potential to greatly reduce the distribution channel from hundreds of miles to a few hundred feet.Online Exclusives Michael Barbella, Managing Editor 12.21.21
-

Patient-Specific Implants Often Require More Testing
Identifying a "worst-case scenario" for testing multiple customizable sizes can be quite challenging for manufacturers.Online Exclusives Michael Barbella, Managing Editor 12.17.21
-

Evolving Regulatory Expectations are Complicating Device Testing
Broad standards can make it difficult for implant manufacturers to fully understand regulatory requirements.Online Exclusives Michael Barbella, Managing Editor 12.14.21
-
Biocompatibility & Testing

AI Tech Requires Customized Clinical Evaluation Strategies
Testing AI’s safety, efficacy, accuracy, cybersecurity, and learning capabilities in the orthopedic arena will be challenging.Online Exclusives Michael Barbella, Managing Editor 12.10.21
-
Biocompatibility & Testing

Testing Today’s Orthopedic Devices: A Roundtable Discussion
Besides performance, orthopedic implants must be assessed for their integration and functionality with other product components.Michael Barbella, Managing Editor 09.14.21
-
Biocompatibility & Testing

Automated Data Analysis: Making the Case
Shifting to automated data analysis for process qualification activities can improve the effort for manufacturers and their customers in several ways.Jack Wieland, Project Manager, Lowell Inc. 09.14.21
-
Biocompatibility & Testing

Disinfectant Efficacy Testing: How Limiting Bioburden Keeps You Compliant
Medical devices are only effective if they can perform their intended functions safely, and safe performance starts with reducing bioburden.Leslie Tavares, Melinda Johnson and Jessica Lawrence, WuXi AppTec 09.14.21
-
Biocompatibility & Testing

Human Factors Usability Testing at Home
Online, remote usability testing at home for orthopedic patients has turned out to be a valuable option.Maria Shepherd, President and Founder, Medi-Vantage 08.17.21
-
Biocompatibility & Testing

Keys to a Successful First Article Inspection
First article inspection is a necessary step to ensure a manufacturing process will create a medical device that works as designed and meets requirements.Jack Wieland, Project Manager, Lowell Inc. 08.17.21
-
Biocompatibility & Testing

Sotera Health Acquires BioScience Laboratories
Strengthens Sotera Health’s antimicrobial and antiviral lab testing capabilities.Charles Sternberg, Assistant Editor 03.11.21
-
Biocompatibility & Testing

Lessons from the Lab: Testing Orthopedic Devices
Changes in testing protocols and revised regulatory demands make testing experts even more valuable than they’ve ever been.Sean Fenske, Editor-in-Chief 09.15.20
-
Biocompatibility & Testing

Nelipak Facility Earns ISO/IEC 17025 Accreditation
Provides a comprehensive range of testing services to support package testing requirements.Nelipak 09.10.20
-
Biocompatibility & Testing

Avoiding Contradictions: Testing Advice for Manufacturers
Manufacturers need to evaluate their approach to preclinical testing. Avoiding contradictory study design is the most effective path forward for study sponsors.Mark R. Cunningham, Ph.D., Senior Director of Science and Technology, Technical Services, WuXi AppTec 08.11.20
-
Biocompatibility & Testing

Conducting Chemical Characterization/Risk Assessment for FDA Submissions
Chemical characterization/risk assessment is a powerful methodology for addressing select biocompatibility endpoints.Robert A. Allen, Ph.D., Senior Associate, Regulatory Affairs, MCRA LLC 05.20.20
-

Trump Signs Bill Replenishing Coronavirus Relief Funding
Legislation provides $484 billion for company payrolls, hospitals, small business loans, and a national testing system.Staff and Wire reports 04.24.20
-
Imaging | Large Joint
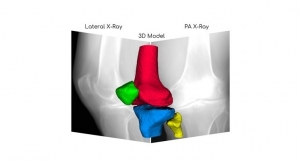
RSIP Vision Launches AI for 3D Knee Image Reconstruction
Creates rich 3D modeling of each knee bone based solely on widely available X-ray images.PR Newswire 03.18.20
-
MIS/Robotics

Rise of the Robots: A Robotic-Assisted Surgery Synopsis
Pre-planning and surgical precision are driving physician acceptance and market adoption of robotic-assisted technology in orthopedics.Michael Barbella, Managing Editor 03.13.20
-
Biologics
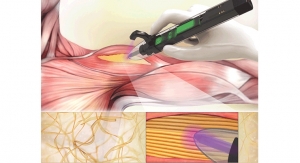
Made-to-Order Muscles Could Be Served in a Hospital Near You
Dr. Ali Tamayol recently developed a handheld bioprinter pen that could significantly change the way musculoskeletal procedures are performed.Sam Brusco, Associate Editor 03.13.20
-
Design
The Importance of Usability/Human Factors Testing
When is the work of human factors over, and how does your team know they are prepared for summative testing?Maria Shepherd, President and Founder, Medi-Vantage 03.13.20
-
Large Joint

Particle Particulars: Why this Analysis Is Foundational for Successful Devices
In the mid-2000s, medical device experts were hopeful they’d found one solution to longer-lasting implants.Dawn A. Lissy, President & Founder, Empirical 03.13.20
-
Design | Instruments

A Case Study of Design Controls and Orthopedic Instruments
Most orthopedic instruments are Class I so manufacturers aren’t required to apply formal design controls, but recently, attitudes are changing.Victoria Trafka, President & Principal Engineer, Engineering & Quality Solutions Inc. 03.13.20
-

Senators Introduce Bill to Address Medical Supply Vulnerabilities
Bill would study and develop action plan to address U.S. dependence on foreign-made medical equipment and boost domestic production and supply.Lamar Alexander (R-TN) and Dick Durbin (D-IL) 03.12.20
-
Supply Chain

Senators Introduce Bill to Address Medical Supply Vulnerabilities
Bill would study and develop action plan to address U.S. dependence on foreign-made medical equipment and boost domestic production and supply.Lamar Alexander (R-TN) and Dick Durbin (D-IL) 03.12.20
-

FDA Suspends Foreign Inspections
The agency has postponed most foreign inspections through April, effective immediately.Food and Drug Administration 03.10.20
-

FDA Suspends Foreign Inspections
The agency has postponed most foreign inspections through April, effective immediately.Food and Drug Administration 03.10.20
-
Spine/Neurology

DiscGenics Completes Enrollment in U.S. Trial for DDD Cell Therapy
Study will evaluate the efficacy of injectable Discogenic Cell therapy in patients with symptomatic, single-level, mild to moderate lumbar DDD.DiscGenics Inc. 03.05.20
-

FDA Offers Coronavirus Update Regarding Product Safety
The agency outlines the steps it will take to ensure the quality of foreign products.U.S. Food and Drug Administration 02.25.20
-

FDA Offers Coronavirus Update Regarding Product Safety
The agency outlines the steps it will take to ensure the quality of foreign products.U.S. Food and Drug Administration 02.25.20
-
Spine/Neurology

Magnet-Controlled Bioelectronic Implant Could Relieve Pain
MagNI can be wirelessly charged and programmed with magnetic fields.Rice University 02.20.20
-

3D Printed Devices in Orthopedic Implants Comprise Nearly Half of Total Sales
Market expected to grow 16 percent annually through 2029.Future Market Insights 02.19.20
-
Packaging & Sterilization
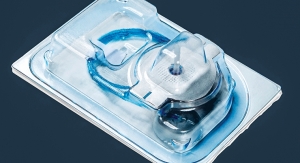
Proceed with Caution: Medtech Seeks Safe Packaging and Sterilization Solutions
Concerns surrounding ethylene oxide have brought the packaging and sterilization segments to the forefront for medical device manufacturers.Mark Crawford, Contributing Writer 02.18.20
-

Recalibrating for 2020: Tips for a Better New Year
In 2019, I had to take a long, hard look at both my businesses and my industry to find balance and a way forward through the ripple effects of big changes.Dawn A. Lissy, President & Founder, Empirical 02.18.20
-

ARCH Acquires MMi Sonora
Bolsters ARCH’s position as a supplier to the medical market.Arch Global Precision 02.18.20












